GIMS.pharma
Was ist GIMS.pharma?
Die Pharmakogenetik untersucht, wie genetische Varianten die Verstoffwechslung von Medikamenten beeinflussen. Jeder Mensch trägt solche Veränderungen in sich. Sind diese bekannt, kann der Arzt das Wissen für eine maßgeschneiderte Medikamententherapie nutzen, um unerwünschte Arzneimittelwirkungen zu vermeiden.
Mit GIMS.pharma kann pharmakogenetisches Expertenwissen einfach im Labor implementiert werden: Die Software ermöglicht eine automatisierte Verarbeitung von pharmakogenetischen Analysedaten zu validierten und gebrauchsfertigen Medikamenten- und Dosisempfehlungen.
GIMS.pharma unterstützt dabei:
- Medikamentensicherheit zu erhöhen
- Wirksamkeit zu optimieren
- unerwünschte Nebenwirkungen und Komplikationen zu vermeiden
- Hospitalisierungsraten und Kosten zu senken
MAIN GOALS
![]()
improve
patient safety
![]()
optimize
drug efficacy
![]()
avoid adverse drug events
and complications
![]()
reduce hospitalization rates
and health care costs
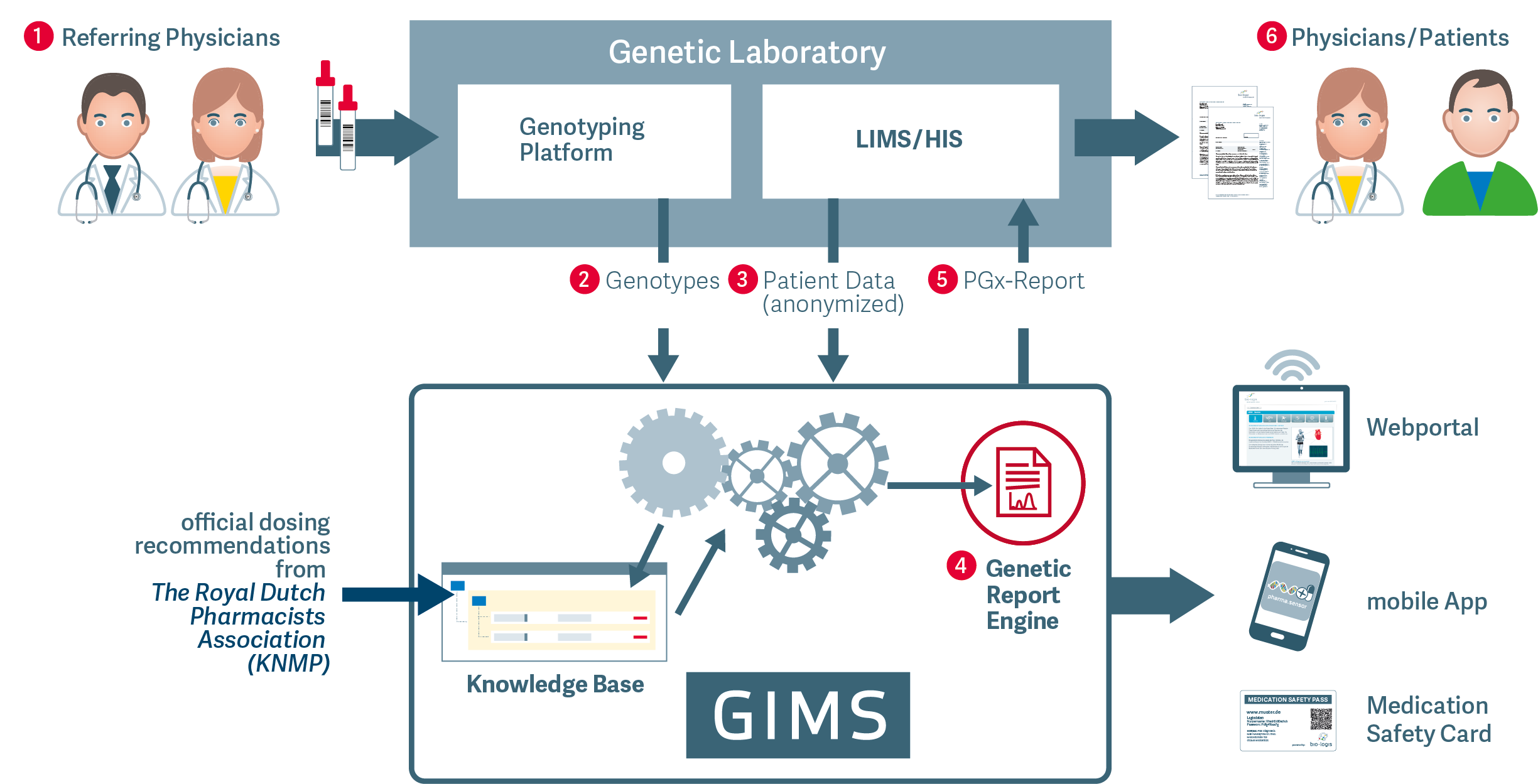
How it works

1. Physicians at Point of Care (PoC) collect blood samples from patients and send it to laboratory
2. Samples processed with any genotyping platform
3. Lab-results transferred via secure VPN-connection into bio.logis GIMS
4. Production of diagnostic reports including validated clinical interpretation and dosing recommendations
5. Diagnostic reports to be transferred into any laboratory or hospital information system for medical validation
6. Validated content provided as documents and via digital channels with smart functions to physicians and patients at point of care
Die wichtigsten Vorteile
SaaS für PGx
- 13 Gene, 49 Genotypen, relevant für 78 Wirkstoffe
- einfache Integration in IT-Infrastrukturen von Kliniken / Laboren
- CE zertifiziertes Medizinprodukt, DSGVO geprüft datenschutzkonform
Digitaler Output
- für Ärzte und Patienten am Point of Care
- in Anwendungen wie Webportal, mobile App, Medication Safety Pass
Contact block
Durch Ausfüllen und Versenden des Kontaktformulars stimmen Sie automatisch der Verarbeitung Ihrer Daten gemäß Artikel 6, Absatz 1a der DSGVO zum Zweck der Beantwortung Ihrer Anfrage zu. Weitere Informationen finden Sie in unserer Datenschutzerklärung.

